








The Impact of Ions On Coffee Flavours





How much can a single molecule impact a cup of coffee?
As a disclaimer, I am not a scientist, and my chemistry background is limited to a high-school major. Most of my research is based on the sensory analysis of different minerals onto the flavours of coffee, using a pragmatic, replicable, predictable and transferable approach – which I believe is paramount to approach this topic and make it accessible to all.
In 2019, after coaching Carlos Escobar in the Western Region Australian Brewers Cup to a first-place finish, I realised that water mineral composition had a massive impact onto coffee. Since then, I decided to deep-dive into water chemistry to better understand how water impacts the flavours of coffee, and I believe my findings have come a long way since.
As part of my work, I taste coffee all around Australia and in many countries around the World. I quickly realised that by controlling and better understanding water chemistry, we could not only get more consistency in cafes but also improve quality and customer experience.
“Nowadays, cafes have access to amazing coffee quality, but the lack of education around water chemistry is the biggest factor that prevents them from getting the most out of it and for consumers to really see the added value.”
In the past 4 years, my team and I have leveraged this deeper understanding about water chemistry to win several local coffee competitions, national championships, and even top finishes in the World Championship. In this article, I will share some of my key learnings when it comes to using water as a tool to improve coffee’s flavours.
From the moment I started researching this topic, I was confronted by 2 issues:
– Most of the information you can find online regarding water chemistry for coffee use abstract scientific concepts such as pH, Alkalinity (KH), total PPM, General Hardness (GH), Temporary Hardness which make it very difficult for anyone to utilise and interpret to improve their coffee.
– Most of the time, people refer to minerals as Magnesium, Calcium or Bicarbonate, however, these are individual ions that can’t be found by themselves and do not offer a full picture of how they impact the flavours of coffee.
For instance, baristas often refer to the TDS of their water. TDS (total dissolved solid) is the concentration of dissolved substances into water. What it doesn’t tell you and what is inside that water. I once heard someone say that it is the same as describing a music band playing talking about the decibel level. It does not tell you what instruments are playing, just if it’s loud or quiet which is irrelevant in most cases (if you water is sitting at 20ppm then it’s likely too soft and if it sits at 500ppm, it’s likely way too hard – regardless of what’s in it).
I have always strived to make educational content accessible to all, mainly by explaining things simply or by using metaphors that anyone can understand.
“If you can’t explain it simply, then you don’t understand it well enough” – A.Einstein.
Keeping this philosophy in mind and without getting too technical, here is what you need to know about chemistry to improve the flavours of your coffee.
Cations and Anions – the perfect balance
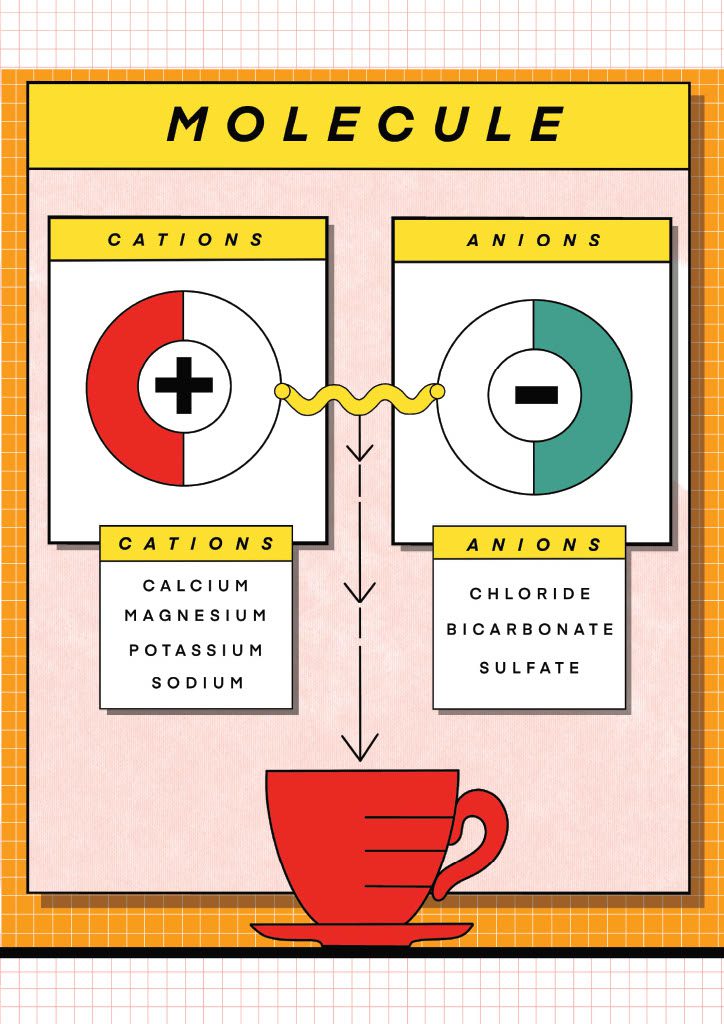
A molecule is a group of two or more atoms bonded together covalently, i.e., by sharing of electrons between them. Generally, a molecule does not have ions in it, as all the atoms in it are electrically neutral. However, some molecules may form ions when dissolved in water or other solvents.
In such cases, the molecule dissociates into charged particles known as ions. For example, when sodium chloride (NaCl) is dissolved in water, it dissociates into positively charged sodium (Na+) and negatively charged chloride (Cl-) ions. We call these positive ions Cations and the negative ones Anions.
Therefore, one molecule of NaCl forms two ions when dissolved in water.
In solid form, these 2 ions can never be separated. It is therefore impossible to find just Magnesium or Calcium or Bicarbonate on their own, as each ion is always bound to another ion of opposite charge. For that reason, when building a water from scratch (adding minerals to a demineralised base) we should always consider the 2 ions of each mineral that we use, as they both impact the cup.
For instance, Magnesium Sulfate is composed of Magnesium (Mg^2+) ions and Sulfate (SO4^2-) ions, but most of the time, people only refer to the Magnesium ion, forgetting entirely about the Anion it’s attached to and how this Anion will taste.
In my research, I have consistently found that certain ions brought very positive attributes to the cup and had a very high ceiling (Magnesium, Calcium, Sodium, Potassium), however, other ions, generally anions can bring positive notes, but they reach their maximum much faster (Sulfate, Chloride, Bicarbonate).
Ions and coffee seasoning
Baristas and brewers can easily be compared to chefs. They work with raw products and transform them into a new finished one. A chef works with a wide variety of ingredients to create a dish, a brewer works with coffee, water and can utilize ions to lift the flavours in the final cup.
Ions impact coffee flavours the same way seasoning and condiments impact the flavours of a dish. They cannot magically turn bad coffee into a delicious one, but they are often the difference between a good and exceptional cup.
I’ve found that maximising the amount of each seasoning/ion always leads to a better dish/cup. In cooking, we always try to maximize each seasoning up until the point it brings more negative attributes than positives.
The analogy does not stop here! Since Cations and Anions are always paired, you cannot find Magnesium or Calcium on their own, as they always come attached to an anion. This means that if we want to use one specific ion, it will always come with another one which isn’t always desirable (each ion has a maximum ceiling depending on the coffee and the expression you have in mind). If these were individual seasoning components, you wouldn’t be able to add salt or black pepper or olive oil only, it would have to be paired with another seasoning like chili, vinegar, nixed herbs.
Adding minerals to coffee is therefore the equivalent to seasoning a dish with a seasoning blend like Chili Salt, Oil and Vinegar, Pepper and Herbs.
If we are using one of those seasoning blends, we need to look at the ceiling of each component, the maximum level until they start bringing negative attributes.
If we decide to use Chili Salt, it is likely that our dish will be too spicy before it becomes too salty. You will find that Chili Salt reaches its limit with Chili, not Salt. This is why it is paramount to consider the 2 ions separately.
Magnesium Sulfate (MgSO4) is a great example where Magnesium has a high ceiling, but Sulfate will quickly make your coffee taste bitter, dry and hoppy. Sulfate would prevent you from adding more MgSo4 that you’d want, but if you still have room to add more Magnesium, you can look at other Anions attached to it, and in comes Chloride.
In the ASCA Brewes Cup Championship, I explained how I blended Magnesium Chloride (MgCl) and MgSo4. This way I could introduce Chloride ions (which bring body and sweetness) and Sulfate ions (floral, citrus notes) in small quantities while maximizing my amount of Magnesium (brightness and fruity notes).
This blending principle is something I applied to all 7 ions that I used in my brewing water.
When building my water recipes, I always look at the maximum amount of each ion that I can use and always try to maximize them until I reach the point of no marginal gain.
How do these ions impact the flavours of coffee?
Research done of the impact of minerals on coffee seem to suggest that only Carbonate ions impact the extraction process. All other ions do not seem to interfere at that stage, that said, all ions have a specific taste and can be detected by our taste buds which will in turn impact the flavours of the final brew (different ionic compositions will impact the way water tastes, which is why different brands of water from the Supermarket taste different, because they have a different “seasoning” composition).
In the final part of my presentation, I explained how my coffee tasted with our filtered water in the office. It showcased notes of fresh strawberry, rose, cherry, citrus, stone fruits, orange blossom and elderflower. In the centre of the Ion Flavour Transformation, I added the ion recipe that I used for this coffee and on the right-hand side, how the coffee then tasted with this new, tailored water.
Note that I had to do minor tweaks to my water recipe on the day of the finals due to unforeseen circumstances.
I put ions together in groups to showcase their combined benefits and impact rather than showcasing them in the pairs them come.
Calcium, Potassium and Chloride together bring a lot of sweetness, a heavier body, a rich coating texture and a delicious confectionary-like quality. They work incredibly well with Naturals or coffees that underwent a long fermentation to bring out more of the red and pink flavours.
Magnesium and Sodium produce a vibrant, sparkling and mouth-watering acidity that highlights tropical notes like pineapple, passionfruit and mango.
Bicarbonates adds structure to the cup, balancing and controlling the acidity. When coupled with Sulfate, in small quantities, it brings out more of the floral qualities in coffees that have floral notes like geisha varieties. This gave me more intense notes of hibiscus, rose and cherry blossom.
Going back to that chef/barista analogy, as a cooking afficionado, I’ve always put more trust into the recommendations of chefs rather than food scientists. Chefs can’t usually explain WHY things happen in a certain why during the cooking process (which is a complex series of chemical reactions) but they know the answer to the WHAT happens by doing xyz. They are mainly focused on the concrete, pragmatic understanding of the impact of individual variables onto the final dish, rather than trying to explain why.
I am a chef, not a food scientist. I do not have the scientific baggage needed to explain why ions impact coffee flavours the way they do, but what I do know is what impact they have on the final cup and what to change to tweak and improve coffee. Moreover, there is contradicting evidence as to whether all ions impact the brewing process or if only Carbonate ions do. I believe this is far more important than trying to explain certain phenomenon with pseudoscience.
I’ve attended countless cuppings where people would judge a coffee and score it down on specific attributes not because of the coffee itself or the roast but because the brewing water did not do justice to this coffee. The feedback then goes onto the roaster/importer/producer which has no control over the way coffee is brewed, and that isn’t fair, and it needs to change.
“There is a tendency in the industry to ask for more and more from the producer to improve flavours in coffee. I believe that by having a better understanding of the impact of ions on coffee and using that as a flavour improvement tool, we can relieve some of that pressure on producers.”
Only few people have witnessed how potent and precise it is to dial-in a coffee only by changing the ion composition of the brew water. This is why we are launching a series of Workshops and Masterclasses to highlight the power of ions and their impact on coffee flavours with the coffee community.
By Coffee Excellence Manager, Simon Gautherin